 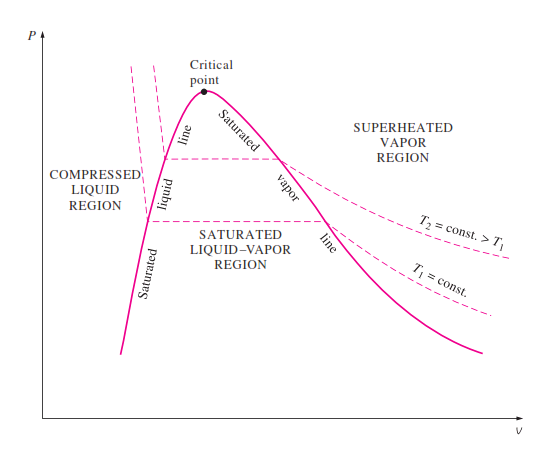  
What can be said about a pure sample of Z’s melting point?Ī: It would have a melting point of between 117☌ and 121☌.ī: It would melt at temperatures lower than 117☌.Ĭ: It would melt at temperatures higher than 121☌.ĭ: Its melting point cannot be determined. ExamplesĪn impure sample of Z melts over a range of temperatures ranging between 117☌ and 121☌. Conversely, the lower the pressure exerted on the liquid, the lower its boiling point would be. The greater the pressure exerted on the liquid, the higher its boiling point would be. Pressure also affects boiling point of a substance. The substance would boil over a range of temperatures.The more impurities a substance contains, the higher its boiling point will be. With impurities, the boiling point of a substance is affected in two ways too: 
Melting would occur over a range of temperatures and not at a constant/fixed temperature.Ī pure liquid has a constant/fixed boiling point.The more impurities a substance contains, the lower its melting point will be. Elements and compounds are both examples of pure substances. Mixtures can be classified as homogeneous or heterogeneous. Mixtures are physical combinations of two or more elements and/or compounds. For example, pure ice melts completely at zero degree Celsius but the ice we make at home from tap water will start to melt before zero degree Celsius. A pure substance is a form of matter that has a constant composition and properties that are constant throughout the sample. With impurities, the melting point of a substance is affected in two ways: it will melt completely at one temperature only. SolidsĪ pure solid has a constant/fixed melting point. A compound is formed when two or more elements combine chemically. To determine if a substance is pure in school laboratories, we can check the substance’s melting or boiling points or use chromatography (see Separation Techniques). A compound is regarded as a pure substance to the mixture is not. Determining if a substance is pure is important because, for example, if a medication consists of impurities, it may cause harm when ingested. In this chapter, we would look into how to differentiate if a substance is pure (or is it a mixture). In contrast, a mixture is a substance which has two or more different substances mixed together (not chemically combined). If, however, the material remains hylotropic and resists fractionation under all available conditions, it is classed as an element.A pure substance consists of only one type of substance not mixed with others. A solution, on the other hand, may remain of constant composition when attempts are made to fractionate it by a single method, for example, by distillation under a given pressure, but generally begins to break up when a second process of fractionation is tried, for example, by fractional distillation under a different pressure, or by freezing. According to these definitions, a chemical compound is merely a ‘hylotropic’ substance which remains constant in composition over a range of temperatures and pressures, within which it resists all attempts at fractionation. TIMMERMANS has chosen as a basis for his monograph on “Chemical Species” the definitions given by Wald in 1897, and used by Ostwald in 1904 in the Faraday Lecture in which he tried to show that the molecular theory had become a superfluous hypothesis in view of the rapid development of the applications of thermodynamics to chemistry. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
